 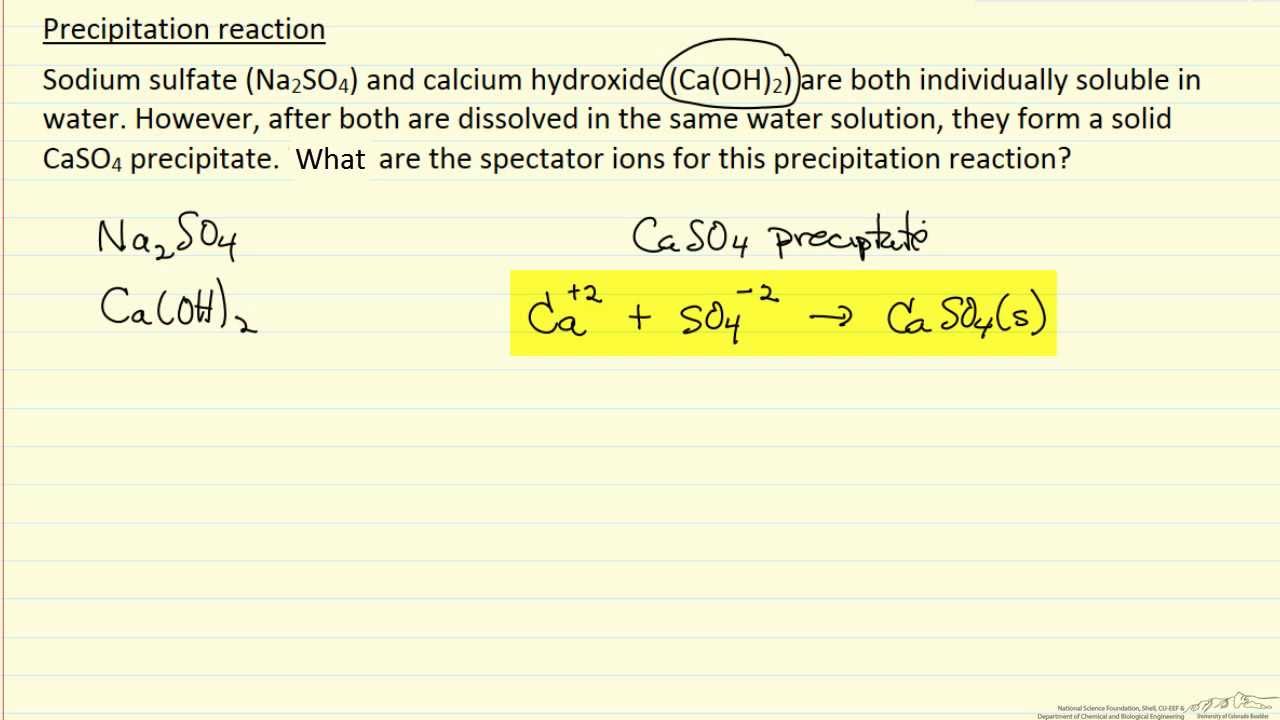
It generally follows a power-law relationship such that Nucleation Rate µs n(2). The nucleation rate often is defined as the number of crystals produced per unit time per unit volume of crystallizer. The numerator is the ionic activity in the supersaturated solution and the denominator represents thermodynamic equilibrium. Where g denotes the activity coefficients for the ions in solution, and are the concentrations of ions in the supersaturated solution, and K sp is the solubility product, which equals at equilibrium at the given solution conditions. For dicalcium phosphate, the supersaturation level can be expressed as: The relationship between nucleation and growth provides the basis for understanding the conditions needed to promote crystal growth. This corresponds to the pK d value of the dissociation constant for that compound. Solubility decreases rapidly above a pH of about 2.5. 2H 2O in water at pH 2-5 and 37☌, as estimated from solubility isotherms reported by Elliott.Figure 1 illustrates the solubility/dissociation behavior of CaHPO 4 Understanding the reaction product’s solubility behavior in the mother liquor is critical to understanding supersaturation, the driving force for crystallization. We focused our efforts on crystallizing CaHPO 4 2H 2O, is the primary product at pH 2-4 and temperatures below 80☌.A review of the literature indicates that hydroxyapatite, Ca 10 (PO 4) 6(OH) 2, is the primary product obtained at alkaline conditions, while dicalcium phosphate dihydrate, CaHPO 4 This depends upon pH and also upon whether the products of the various possible calcium phosphate reactions are fairly soluble and able to undergo further reaction in solution, or only sparingly soluble. The mix of calcium phosphate reaction products generated depends upon the ion speciation - that is, the type of ions present in solution, whether Ca 2+, H 2PO 4-, HPO 4 2-, or PO 4 3-, among others. This article explains why and illustrates the importance of understanding and controlling solute supersaturation levels in a crystallization operation. The low product solubility not only allowed removal of more than 95% of the phosphate present in the feed, but also made the task of building large, filterable particles challenging. Because calcium phosphate solids are only sparingly soluble in water at neutral pH, the filtrate from this process could be sent to the WWTP. The processing scheme involved adding slaked lime to a batch of phosphoric acid in a stirred tank, and filtration of calcium phosphate solids from the resulting slurry we opted for batch operation as it fit well into the specialty chemicals plant where the phosphoric acid waste was generated.  
Incineration methods are sometimes used, but they can be expensive and generally do not allow recovery of the phosphate content.Ī number of years ago we implemented an alternative method involving reaction of phosphoric acid with hydrated or “slaked” lime (Ca(OH) 2 particles slurried in water) to precipitate calcium phosphate solids. In disposing of phosphoric acid waste, simply neutralizing the waste with caustic and sending it to a wastewater treatment plant (WWTP) often is not an option because the resulting phosphate salts are soluble in water and the total amount of phosphate that can be discharged from the WWTP is strictly controlled for environmental reasons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
